What’s the Difference Between Quality Assurance and Regulatory Affairs?
If you’re navigating compliance in a small or early-stage medical device company, chances are you’ve come across two essential terms: Quality Assurance (QA) and Regulatory Affairs (RA). These functions are both crucial to getting a safe, effective product to market, but what exactly do they do and how are they different?
The Rule of Thumb: Internal vs. External
One of the clearest ways to distinguish QA from RA is this:
-
- Regulatory Affairs (RA) = External: They interact with regulators, manage submissions, and ensure the company meets legal requirements for product approval and market access.
- Quality Assurance (QA) = Internal: They build and maintain the quality system, guide internal processes, and make sure the organization is actually doing what it’s supposed to do.
Both functions are about compliance, but they focus on different elements of it.
What Does Regulatory Affairs Do?
RA is responsible for:
-
- Understanding and interpreting regulations regarding product claims (e.g., FDA, EU MDR, Health Canada).
- Preparing and submitting regulatory filings: 510(k)s, PMAs, Technical Files, etc.
- Managing interactions with regulators and Notified Bodies.
- Keeping track of regulatory changes and ensuring continued compliance post-market.
- RA spans the product lifecycle from R&D through clinical stages, manufacturing, and post-market surveillance
They are the bridge between your company and external authorities, and often the first to know when regulatory expectations shift.
What About Quality Assurance?
Quality Assurance is the function that ensures products are consistently made and controlled according to standards. This includes design and development of the product, especially in smaller companies, where QA is deeply involved in:
-
- Defining and documenting how things get done; from design controls to manufacturing to post-market feedback.
- Collaborating with teams to develop SOPs, batch records, risk management files, and more.
- Building and maintaining the Quality Management System (QMS) (e.g., under ISO 13485 or 21 CFR Part 820).
- Leading internal audits, CAPA processes, and quality planning.
RA might say, “Here’s what the regulations require us to do to get approval.” QA might respond, “Here’s how we’ll do that reliably, every time in our real-world operations.
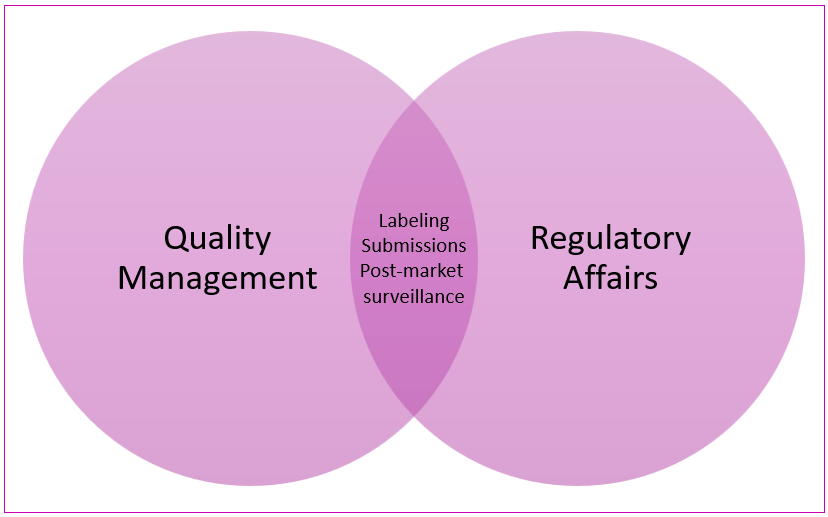
The Blurry Line in Practice
In a startup, the lines between QA and RA often blur. It’s not unusual for one person to be both the regulatory lead and the quality system owner. Even in larger companies, the handoff between RA and QA isn’t always clear-cut. For example:
-
- Who writes the labeling content? RA.
- Who ensures the labeling is accurate, controlled, and traceable? QA.
And while QA is typically internal-facing, it doesn’t mean QA never interacts with regulators, especially during audits and inspections.
At a small or early-stage company, RA may be needed infrequently, in small bursts, such as when a new product is under development, or when a product will launch in a country where it has not previously been sold. But QA is usually involved in day-to-day operations at a steady pace. QA may have peaks of work around product submissions or launches too, but they are also engaged in routine operations.
QA Strategy is Not Just Checking Boxes
There’s a misconception that QA is just about batch record reviews or procedural compliance. But effective quality management, at the strategic level, involves:
-
- Planning how and when quality is built into the product lifecycle.
- Collaborating with R&D, operations, and suppliers to define requirements up front.
- Enabling scalable, compliant processes that evolve as the company grows.
In the end, both QA and RA are essential to building a compliant, resilient, and trustworthy medical device company.
Understanding the distinction
-
- QA as the operational backbone ensuring internal processes work
- RA as the outward-facing expert navigating regulatory landscapes
helps small teams divide responsibilities more clearly and build smarter from the start. Whether you’re wearing one hat or both, recognizing the complementary nature of these roles is key to getting your product to market – and keeping it there.
Need help balancing quality and regulatory work? We support startups in building practical, right-sized systems.

